Understanding the long-term carbon-cycle: Weathering of rocks – carbon-sinks
By John Mason / SkepticalScience: Because of its length, I’ve broken it up into bookmarked sections for easy reference: to come back here click on ‘back to contents’ in each instance.
Contents
Introduction: what is weathering?
Carbon dioxide and rock weathering: the chemistry
Limitations to the precipitation of calcium carbonate: the Carbonate Compensation Depth
The significance of weathering as a carbon-sink
Deep weathering of rocks: an illustrated example from Mid-Wales, UK
How breaking up minerals affects their weathering-rate: mountain-building as an accelerant
Picking up signals of major weathering episodes in the geological record
Introduction: what is weathering?
Weathering is a familiar process to us all. It involves the chemical reactions between chemical compounds in the atmosphere and chemical compounds on the planet’s surface. When your car’s exhaust pipe falls apart noisily, it is because the steel from which it was constructed has, over several years, reacted with oxygen and rainwater to form rust. It has weathered. But that’s a relatively fast example involving a relatively unstable compound. The compounds making up the vast majority of Earth’s land surface – the minerals that make up rocks – are, by and large, very slow to react. As a consequence, large-scale weathering is a process that takes place on a timescale of millions of years, over which periods it constitutes a critically important carbon-sink.
Why a carbon-sink? Because, via weathering of rocks and reprecipitation of weathering products as carbonate sediments (e.g. limestones), huge quantities of atmospheric carbon dioxide end up locked away for a very, very long time. The process begins when CO2 dissolves in droplets of water, up there in the clouds. The resulting solution, which reaches the surface as rainwater, is weakly acidic:

CO2 + H2O = H2CO3
(or carbonic acid – the old name for carbon dioxide was carbonic acid gas)
Rainwater containing carbonic acid is able to react with most minerals at varying rates according to their chemical stability. Now, some naturally-occurring minerals are extremely stable. Think about gold, eroded mechanically from ore deposits and then recovered by prospectors, maybe hundreds of thousands of years later, from river-gravels by panning. Or quartz (silicon dioxide), found as hard, white pebbles on beaches. They’re both pretty bomb-proof.
At the other end of the scale are the very unstable minerals, such as sulphides, compounds of various metals with sulphur. Sulphides readily weather in the surface or near-surface environment, so that most ore-deposits have near-surface zones in which the products of primary sulphide weathering – the secondary minerals – are to be found. The copper carbonates, azurite and malachite, are examples of secondary minerals that will be familiar to most readers. Such minerals can occur in large quantities where the weathering has been prolonged – over a few million years – but the relative rapidity of the process is demonstrated by the fact that thin films of often colourful secondary minerals are a common sight along the walls of mine-tunnels only a century or two old.

above: primary (left) and secondary (right) copper minerals. The common primary ore, chalcopyrite, is a sulphide of copper and iron that reacts readily with air and moisture at the surface. Prolonged weathering forms colourful secondary minerals in large quantities, like the blue and green copper carbonates, azurite and malachite (the illustrated specimen is about 6 inches long). These copper carbonates constitute a localised, and therefore relatively minor, long-term carbon-sink, compared to the very widespread occurrence of limestones. Photos: author
In between these extremes, from the bomb-proof to the downright unstable, there exists a whole spectrum of mineral stabilities, but an important point is that most of the numerous mineral species that make up rocks lie towards the stable end of that spectrum: whilst they do react with carbonic acid, they do so at a very slow rate, on geological rather than human timescales. So let’s now take a look at what happens.
Carbon dioxide and rock weathering: the chemistry
When carbon dioxide dissolves in droplets of water to form carbonic acid, it dissociates (splits into reactive charged particles or ions):
H2CO3 ⇌ HCO3− + H+
The two ions that occur as a result of this process are the bicarbonate ion, HCO3− and the hydrogen ion, H+. Put simply, hydrogen ion activity within a solution is a measure of its acidity: the more active hydrogen ions in a solution, the more acidic it is and the better at dissolving other compounds. The ‘H’ in pH – the scale by which acidity is measured – is hydrogen. The pH value of 7 (pure water) is neutral, values of 7 up to 14 are increasingly alkaline and values of 7 down to 0 are increasingly acidic.
Carbonic acid is a weak acid: ordinary rain thus has a pH of around 5 to 5.5, but there is lots of it available in the environment, so that over prolonged timescales it does a lot of weathering. Other acids may also be present: for example, sulphur dioxide, outgassed during volcanic eruptions or released by industry, likewise dissolves in droplets of water in the atmosphere to produce sulphuric acid, hence the term ‘acid rain’, with a pH of around 4 to 4.5. However, over geological timescales things like major eruptions and human industry are mere punctuation marks in a long sentence.
Rocks that contain carbonates – limestone and dolomite being common examples – react rather more quickly too because the minerals they are largely made from, such as calcite (calcium carbonate) are more reactive than silicates. Carbonate dissolution leads to the formation of karstic landscapes complete with cave-systems lined with deposits of reprecipitated calcium carbonate forming stalactites and other features. The dissolution reaction goes as follows:
H2CO3 + CaCO3 = Ca(HCO3)2
carbonic acid + calcium carbonate = calcium bicarbonate (in solution)
Silicate minerals occur widely in nature. Some globally-abundant rocks – such as basalts – are mostly composed of silicates. There exists a great variety of different silicate minerals – for example the feldspars, micas, olivines, pyroxenes and amphiboles to name but a few groups – that combine silicon and oxygen with potassium, sodium, calcium, magnesium, aluminium, iron and many other elements. Silicates weather via rather more complex reactions, but let’s simplify things with a generalised equation for the process using the calcium silicate CaSiO3, which occurs naturally as the mineral wollastonite:
2CO2 + 3H2O + CaSiO3 = Ca2++ 2HCO3– + H4SiO4
carbon dioxide + water + calcium silicate = calcium ions + bicarbonate ions + silicic acid (in solution)
The dissolved calcium and bicarbonate ions travel in groundwater to the rivers and thereby find their way to the sea, where they are reprecipitated as calcium carbonate. The reprecipitation is mainly biogenic – it involves various creatures making their shells or skeletons from calcium carbonate:
Ca2++ 2HCO3– = CaCO3 + CO2 + H2O
calcium ions + bicarbonate ions = calcium carbonate + carbon dioxide + water

above: a hand-sized cut slab of limestone, dating from early Carboniferous times (~340 million years ago), with fossil exoskeletons of ancient corals, consisting of calcium carbonate obtained by the organisms from the seawater back at that time. A classic example of an ancient and long-term carbon-sink. Photo: author.
The silica from the silicic acid is used similarly by organisms such as the abundant planktonic diatoms:
H4SiO4 = SiO2 + 2H2O
silicic acid = silicon dioxide + water
Significantly, via the calcium carbonate-generating part of the chemical process, there is a net loss of readily-mobile carbon: we start with twice as much as we end up with. That’s why weathering of silicates is so important. The missing carbon is locked up in the calcium carbonate in the following way: as shelled creatures die and their remains accumulate, we get a carbonate-rich sediment. As the sediments continue to accumulate, our carbonate-rich layer will be progressively buried under new layers of sediment and in time it will turn into solid rock – limestone. Globally, limestones are very common and often occur in great thicknesses (think of the White Cliffs of Dover, for example). That carbon is thereby locked away and immobilised for a very long time. The overall process may be expressed as follows:
CO2 + CaSiO3 = CaCO3 + SiO2
carbon dioxide + calcium silicate = calcium carbonate + silicon dioxide
The basis of the equation written above has been understood since the middle of the 19th Century. A more complex version could be written involving magnesium too: that element can be coprecipitated with calcium to form dolomite, calcium magnesium carbonate with the formula CaMg(CO3)2. Because of their natural abundance and their chemical properties, calcium and magnesium are the two vital elements in this long-term carbonsink. It is important here to point out that weathering of silicates containing e.g. only potassium or sodium does not trap carbon in the same way, because the carbonates of these elements are water-soluble so they do not precipitate to form sediments. Calcium and magnesium are the key.

Limitations to the precipitation of calcium carbonate: the Carbonate Compensation Depth
An important factor regarding the precipitation (or otherwise) of calcium carbonate in the oceans is the Carbonate Compensation Depth (CCD). The reaction:
Ca2++ 2HCO3– = CaCO3 + CO2 + H2O
is reversible: if the physical conditions favour precipitation over dissolution then you will get carbonate precipitates accumulating. But if calcium carbonate dissolution is happening at a faster rate than calcium carbonate precipitation, there is no net gain in solid calcium carbonate: instead calcium carbonate that is already present in solid form starts to dissolve.
What are favourable physical conditions for precipitation or dissolution? Calcium carbonate is more soluble in water at a) lower temperatures, b) at higher pressures and c) in the presence of increased dissolved CO2. These conditions are all satisfied in the world’s deeper oceans, at depths below ca. 5000m, which in broad terms is the Carbonate Compensation Depth. Sediments on ocean floors below this depth contain no calcium carbonate. As the image below shows, such depths are not uncommon.
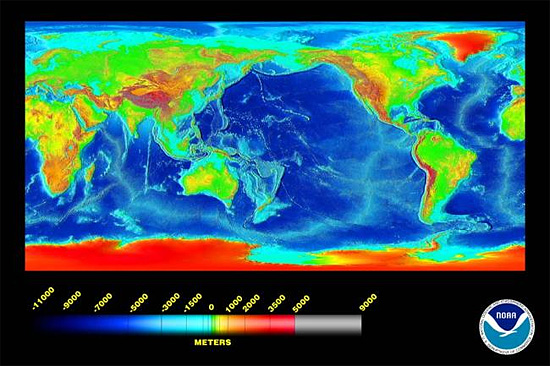
above: ocean depths worldwide. Many of the major ocean basins are deeper than the Carbonate Compensation Depth at ~5000m. Graphic – NOAA.
There is a slight complication to this because, like many substances, calcium carbonate occurs in nature as two minerals – polymorphs – that have different crystal structures. These are calcite (trigonal) and aragonite (orthorhombic). Aragonite is also precipitated in the oceans – molluscs use it to build their shells and corals their endoskeletons – but it has a much shallower compensation depth which varies from less than 1000m in some low latitudes to over 3000m in the North Atlantic.
One effect of increasing dissolved CO2 in oceanic water (ocean acidification) is to lessen the compensation depths of both calcite and aragonite. Whilst such a shift places a further limit to marine carbon sequestration, it also impacts severely on the marine ecosystem. Organisms that need to precipitate aragonite in order to build their skeletons or shells will clearly find themselves in dire straits if the aragonite compensation depth shifts upwards, so that their habitat now lies beneath it. Such a point applies particularly to fixed or benthicfaunas that dwell on the sea-bed because such communities cannot simply up sticks and move: hence they are particularly vulnerable to rapid changes in the chemistry of our oceanic waters.
The significance of weathering as a carbon-sink
The weathering of rocks is estimated to involve the drawdown of about a gigaton of atmospheric carbon dioxide a year. That sounds a bit hopeless when compared to the ~30 gigatons emitted by humans burning fossil fuels every year. Over geological timespans, however, the amount of carbon dioxide removed from the atmosphere via weathering is huge, at around a million gigatons per million years. Globally, limestones and other carbonate-based sedimentary rocks are a phenomenally important carbon sinkthat is relatively stable in nature: they are estimated to hold over 60 million gigatons of carbon – compared e.g. to the estimated total of 720 gigatons carbon dioxide that is present in the atmosphere and the 38,400 gigatons present in all of the oceans.
Limestones do not give up their carbon again readily. The key processes by which they may yield it up include metamorphism (recrystallisation under heat and pressure during mountain-building episodes), subduction (being forced down into the Mantle at a tectonic plate boundary, where the carbon is liberated during melting, leading to CO2 making its way towards the surface in magmas and being outgassed by volcanoes) or industry (people heating limestone to high temperatures in order to make cement). However, compared to human industry, the processes of metamorphism, subduction and volcanism operate over geological timescales, so whilst there is a flux of carbon dioxide going into weathering and thence to limestones, there is also an outward flux going back into theatmosphere. Just as well: it is thought that with no geological resupply, and even with ocean outgassing, the weathering process would remove all atmospheric carbon dioxidein less than half a million years. Just as a rapidly-forming excess of the gas is very bad news, losing it all like that would be disastrous. The long-term carbon cycle makes sure that this isn’t going to happen!
Deep weathering of rocks: an illustrated example from Mid-Wales, UK
On a year-to-year basis, the process of rock-weathering is a slow one. How slow? Well here in Mid-Wales, where I am based, we have a useful example of weathered and unweathered rocks outcropping at surface that demonstrate the time factor as well as anything.
A short crash-course into the geology of Mid-Wales is needed to get this story into context. The rocks of the district consist of slate-grey mudstones, siltstones and sandstones, which were originally deposited as muds, silts and sands on an ancient seabed in late Ordovician and early Silurian times, some 445-435 million years ago. They were then uplifted and folded during a phase of mountain-building and the area has probably remained land ever since. There – done and dusted.

above: slate-grey, relatively unweathered late Ordovician mudstone, typical of the rocks of Mid-Wales. Photo: author
Now we come to the weathering. We know that these mudstones, siltstones and sandstones underwent massive-scale weathering. Why? Because, at scattered localities here and there in the Mid-Wales hills, we find remnants of the weathered rocks. Instead of the normal, slate-grey colour, rocks at such localities are instead various pale pinkish or buff shades. It’s not just a surface thing either: if you whack a piece open with a hammer, you will see that it pervades right through.

above: a broken boulder of pervasively weathered Silurian mudstone from Mid-Wales. The slate-grey colour has been changed to pale pinkish and buff shades. Photo: author
Why are there only remnants of such rocks left behind? The answer is almost certainly glaciation. Glacial ice is a remarkably efficient agent of mechanical erosion. The series of glaciations that took place between 2.5 million and 12,000 years ago fashioned the modern Welsh landscape. Ice-caps over a kilometre thick and valley-glaciers carving their way down to the lowlands are estimated to have eroded away tens to hundreds of metres of strata. As a consequence, in most places in Mid-Wales, the rocks outcropping at surface are the normal, unweathered slate-grey colour, the glaciers having eroded away the deep-weathered stuff. In the 12,000 years since the end of the last glaciation, these rocks have remained visibly unweathered – there hasn’t been enough time for the slow chemical reactions to have had anything more than superficial effects.
How breaking up minerals affects their weathering-rate: mountain-building as an accelerant
In the same area of Wales, the rocks contain numerous mineral-lodes that in the 17th-19th Centuries supported a respectably-sized metal mining industry, with lead, silver, copper and zinc being produced. As with the rocks of the area, the deep-weathered zones of these ore-deposits have (barring a few notable exceptions) been eroded away during glaciation and metal sulphides can often be found quite close to the surface. The old mines neatly demonstrate another critical principle concerning weathering which is that the greater the surface area available to weathering agents, the greater the amount of weathering that can take place in a given time.
Let’s visualise this first with a simple example: imagine a cube of something nice and reactive – iron, for example, 10 centimetres on edge. To calculate its surface area, we first obtain the area of one face: 10cm x 10cm = 100cm2. As a cube has six identical sides, we multiply the area of one face by six, thus getting 600cm2 of iron waiting to react with moist air and go rusty.
Now let’s saw the cube up into 1cm cubes (OK, before anyone says, there’ll be some wastage from the saw-cuts but let’s forget about that). The cube’s volume is 10 x 10 x 10 = 1000cm3, so once the saw has done its work, we have a thousand 1cm cubes. Each will have a surface area of 1 x 1 x 6 = 6cm2. The total surface area of all the cubes will be 6 x 1000 = 6000cm2. By dividing the original cube up into all these little ones, we have increased the surface area available to react by a whole order of magnitude. Cut those centimetre cubes into millimetre cubes and we now have 60,000cm2 of surface available to react, and so on.
Now let’s see the process demonstrated in those mines. When they were being worked, mostly between 100 and 200 years ago, tunnels were driven into the ore deposit and the ore was blasted into small fragments for removal. Invariably, quantities of debris, rich in freshly broken sulphides, were left behind when the mines were abandoned. Both air and moisture then had access to a much larger surface area of reactive sulphides to get to work on. The result was that in just a century or two, the debris became coated or even cemented together by secondary minerals. It’s a good example of a major uptick in the weathering-rate caused by surface area changes. The wide range of secondary minerals present in such coatings and cements (mostly complex sulphates) is indicative of an unstable and immature chemical system that is constantly evolving.

above: small amounts of recently-formed secondary minerals, the result of a few centuries of weathering of freshly-broken sulphides in the metal mines of Mid-Wales. (L): sky-blue rosettes (~2mm across) of the copper zinc carbonate-sulphate schulenbergite with deep blue crystals of the lead copper sulphate linarite. (R): bottle-green crystals of the copper zinc sulphate, ramsbeckite. Photos: D.I. Green
Now, let’s apply the same principles to rocks in general. What processes in nature break up rocks to create a massively increased surface area? Many, but let’s think about the really obvious ones: volcanic eruptions and mountain-building episodes. Both build elevated piles of rock that are prone to collapse by the actions of gravity, earthquakes, rainstorms, explosions in the case of volcanoes and frost action in the case of mountain ranges. In each case, there is a big increase in the surface area of rock available to weathering agents.
Weathering may not, however, take place to any great extent in a high mountain-range like the Himalayas. Temperature is a critical factor: when a landmass is uplifted into a mountain range it will enter a colder climatic zone due to the increase in altitude. Although frost can force rock faces apart and cause rockfalls making a greater surface area of rock available to weather, if it is so cold that there is little or no liquid water available, weathering is inhibited.
The most intense chemical weathering of silicate-bearing rocks instead occurs where temperatures are well above freezing for prolonged periods and rainfall is high, conditions that are especially satisfied in the Tropics – where deeply weathered profiles can extend down from the ground surface to well over a hundred metres depth. Higher temperatures speed up the rates of the chemical reactions and higher rainfall delivers more weathering agents per annum.
Combine such parameters and the answer to the riddle of why mountain-building maylead to enhanced carbon dioxide drawdown becomes clear. Mountain-building episodes facilitate the creation of a vast supply of fresh-broken rock. That rock is constantly being transported away from their cold heights, firstly via gravity (rockfalls), then by glaciers and finally by rivers to a warmer, lowland climate, where the weathering takes place. The Himalayan foothills and the Ganges basin are a good example of such areas where enhanced weathering of rock debris, transported from the mountains and deposited as fine-grained sediment, occurs.
Picking up signals of major weathering episodes in the geological record
Mountain-building episodes occur due to continental collisions that are driven by the slow process of plate tectonics and they typically take place on timescales of millions or tens of millions of years – far more slowly than, for example, the Milankovitch cycles that swing Earth in and out of warm or cool periods on scales of tens to hundreds of thousands of years. For this reason, perturbations to the rate of weathering – and thereby the flux ofcarbon dioxide being consumed by weathering – typically occur slowly, although they may be quite profound, to the extent that significant past changes to climate may have, at least in part, been due to them.
Basaltic weathering is a particularly strong candidate in this respect – basalts are rich in calcium-bearing silicates like plagioclase feldspar and magnesium-bearing silicates like olivine. A major period of basaltic eruption (with much attendant carbon dioxideoutgassing) followed by prolonged intense weathering could potentially create a corresponding peak and dip in atmospheric carbon dioxide levels as outgassing falls and weathering-related carbon dioxide drawdown rises. Can such weathering episodes be detected in the geological past? The answer is quite possibly yes, by studying the ratios of isotopes of the alkaline earth metal, strontium, part of the same group of elements as calcium and able to substitute for calcium in minerals such as calcite (in limestones) and feldspars (in basalts, for example).
Strontium has four stable, naturally occurring isotopes as follows (abundances in brackets): 84Sr (0.56%), 86Sr (9.86%), 87Sr (7.0%) and 88Sr (82.58%). Of these, 87Sr is radiogenic, which means that it has been formed by radioactive decay of an unstable isotope of another element, in this case rubidium 87. The ratio of 87Sr and 86Sr is the one used in the context of weathering: in igneous rocks like basalt it is typically around 0.704 but in other rock-types it is a little higher. Therefore, a period of intense weathering of basalts ought to show up in the isotope record: weathering feldspars release Sr2+ ions in solution which, like the calcium ions, are reprecipitated in limestones, which should then show lower-than-usual 87Sr/86Sr ratios.
This is work-in-progress, but results are promising, taking into account parameters including signals of major erosion, which manifest themselves as upticks in the rates of sediment deposition, and the different weathering-rates of different rock-types. The modelling of fluctuations in the carbon and sulphur cycles for the whole of the Phanerozoic, taking weathering episodes into account, gives plots like the following:
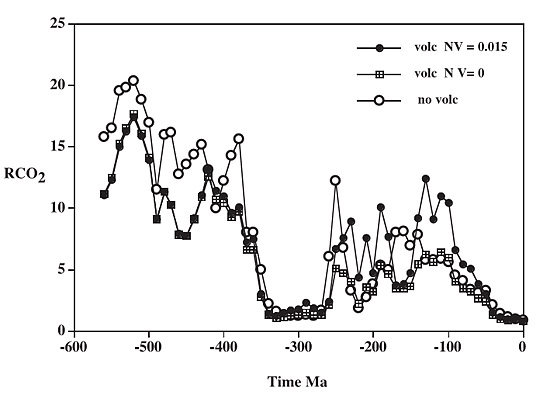
above: ratio of mass of atmospheric CO2 (RCO2) at a past time to that at the pre-human present, calculated via the GEOCARBSULF model, with (volc) and without (no volc) volcanic rock weathering. From Berner (2006).
Several things stand out in this plot, of which the most drastic is the precipitous fall in CO2levels beginning around 375 million years ago during the Devonian, this marking the wide colonisation of the land by photosynthetic plants. It marks an important divide in atmospheric chemistry between a land-plant poor (or plantless, before the Ordovician) world and the richly vegetated world that has existed ever since. The decline in carbon dioxide levels from the Hothouse of the late Mesozoic and early Cenozoic to the cooler world of the past 15 million years is apparent at the RHS of the plot. Older glaciations – the Karoo at 360-260 million years ago and the Andean-Saharan at 460-430 million years ago, are both marked by sharp declines in CO2.
That atmospheric carbon dioxide acts as a key driver with respect to global temperatures is without doubt: it is the detailed reconstruction of atmospheric chemistry that becomes more difficult the further back in geological time one goes. By getting a sound grasp on major, unusually intense episodes of past silicate weathering, another piece of the jigsaw will go into place and those reconstructions will continue to improve.
“The climate has always changed” has been a favourite arm-wave of the contrarian brigade for many years. What they don’t tell you is why, by how much and what were the consequences. In some cases, the consequences were dire, including some of the greatest mass-extinctions this planet has experienced. Causation and magnitude of these events is a major field of research, but perturbations to the long-term carbon cycle, including major outgassing and weathering episodes, are an integral part of the overall picture and may be, in some instances, a prime cause. That we are causing one such major perturbation right now is an indication of the footprint we ourselves are going to leave in the geological record – and given our current track-record, it is not going to be something to be proud of. Indeed, I wonder what the rocks might tell, say 200 million years from now? Perhaps readers might make that the focus of the discussion!
We’ll return to some of these disastrous climatic changes of the past in future posts.
Further reading:
- For a good in-depth discussion of weathering, the following book is well worth finding: Berner, R. A., 2004, The Phanerozoic Carbon Cycle: CO2 and O2: Oxford, Oxford University Press, 150 p.
- The figure above was taken from: Berner, R.A. 2006: Inclusion of the Weathering of Volcanic Rocks in the GEOCARBSULF Model. American Journal of Science, May 2006 vol. 306 no. 5 295-302.
About the Author: EARTH CLIMATE
COMMENTS
- Eric Rignot: Sea level rise there is a distinct possibility it could go faster | Earth Climate on Geological fingerprint suggests rapid glacier retreat
- Eric Rignot: Sea level rise there is a distinct possibility it could go faster | Earth Climate on Eric Rignot: Observations suggest that ice sheets and glaciers can change faster, sooner and in a stronger way than anticipated
- The risk with the path to a hothouse Earth | Climate State on Climate Tipping Points Existential Threat to Our Life Support Systems
- Robert Schreib on Electricity generation prices may increase by as much as 50% if only based on coal and gas
- Robert Schreib on China made a historic commitment to reduce its emissions of greenhouse gases
Support
Paypal DONATE – Your donation goes towards supporting this website, including covering hosting, posting new content, creation of videos, software licenses, or paying invited guest authors. Another way to support Earth Climate is by becoming a Patreon.
